Institut Charles Sadron News

Publié le 14/03/2026 par Drenckhan Wiebke
Foams are everywhere in modern life. They insulate our homes and refrigerators, protect goods during transport, and reduce noise and vibration. Today, most of these foams are made from polyurethane, a highly efficient material — but one that relies on isocyanates, chemicals that are extremely toxic, hazardous to workers, and increasingly restricted by environmental and health regulations in Europe and beyond.
In a new study, researchers from the Institut Charles Sadron (CNRS, University of Strasbourg) and their industrial partner SOPREMA report a promising alternative: a way to produce rigid polymer foams without isocyanates, using a chemistry that is simpler and potentially more sustainable.
A different chemistry for making foams
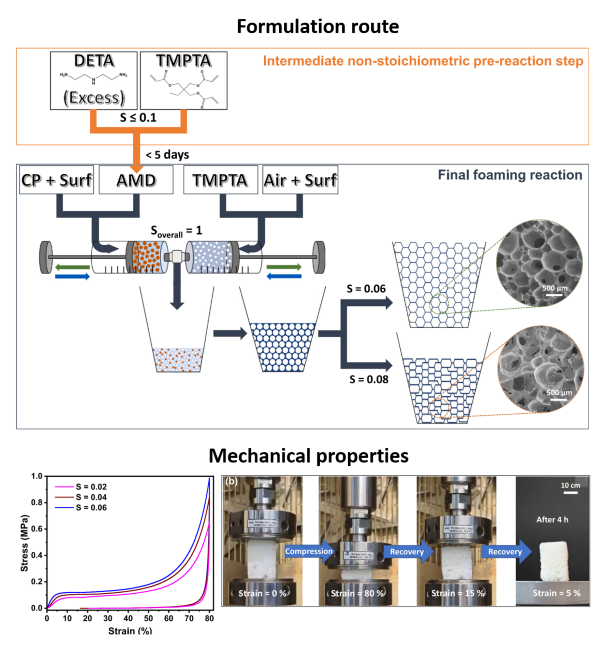
Instead of the conventional polyurethane reaction, the team uses a reaction known as aza-Michael addition to generate rigid polymer foams. This reaction, discovered more than a century ago, allows molecules containing amine and acrylate functional groups to link together under mild conditions, without solvents, catalysts, or toxic by-products, achieving 100% atom economy and satisfying several green chemistry principles.
However, making a good foam is not just about choosing the right chemistry. Foaming is a delicate balancing act: the material must release enough heat to vaporize a blowing agent (which creates the foam bubbles), while solidifying at just the right speed to trap those bubbles before the foam collapses. Achieving this balance without isocyanates has been a major challenge so far.
Controlling foam growth by “stoichiometric engineering”
Beyond the use of aza-Michael chemistry, the key innovation of this work lies in how the reaction is carefully staged and tuned. The researchers introduce an intermediate step before foaming, during which the chemical components are deliberately mixed in a non-perfect ratio. This produces intermediate molecules that act as built-in “reaction moderators.”
By adjusting this ratio — a strategy the authors call stoichiometric engineering — the team can finely control how fast the final reaction proceeds and how much heat it releases, without changing the overall formulation. This level of control allows them to tune the foam’s rise, density, and internal structure simply by adjusting the chemistry beforehand.
Using this approach, the researchers produced rigid, mostly open-cell foams with low densities (as low as ~70 kg/m³), good mechanical strength, and promising thermal insulation performance. Some foams recovered up to 95% of their shape after being compressed by 80%, showing an unusual combination of rigidity and resilience.
Why this matters
This work demonstrates that high-performance foams do not have to rely on isocyanates. By showing that foam structure and properties can be controlled through reaction design rather than complex formulations, the study opens a new pathway towards safer insulation materials.
While the thermal insulation performance does not yet match that of the best commercial polyurethane foams, the authors emphasize that the system is still at an early stage. With further optimization — for example by improving bubble stabilization or creating closed-cell structures — aza-Michael foams could become realistic alternatives for industrial applications.
At a time when regulations are tightening and industries are actively searching for safer material alternatives, this research highlights how fundamental chemistry and process engineering can work together to redesign everyday materials from the ground up.
For more information: the CNRS News and the associated scientific article